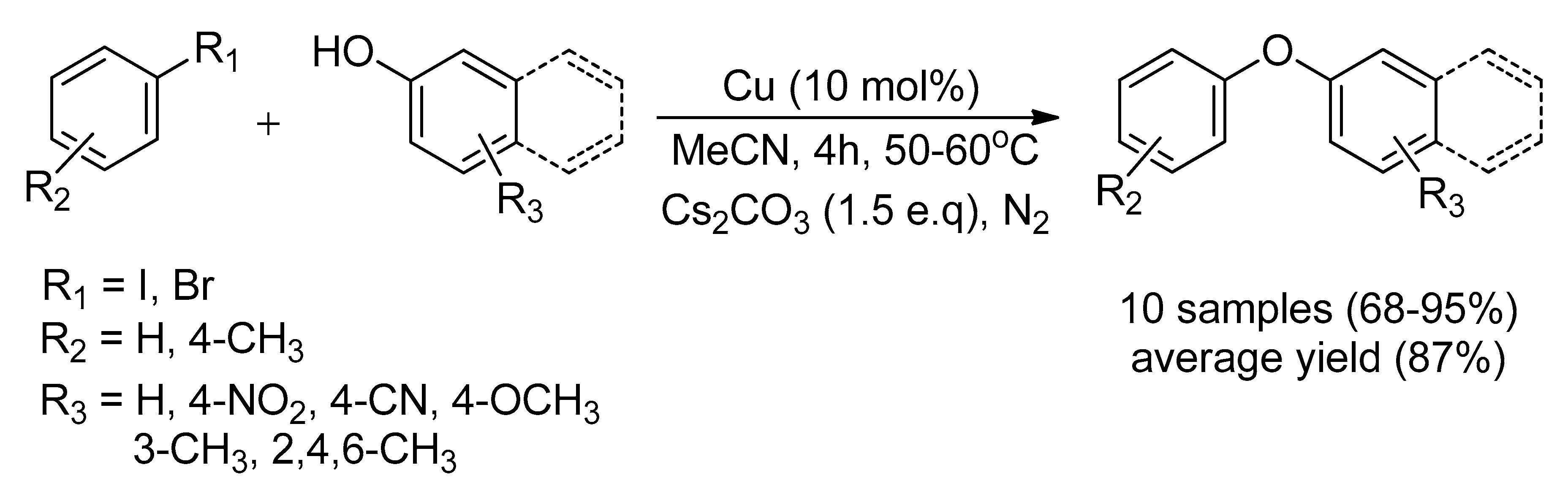
Catalysts | Free Full-Text | Recent Advancement of Ullmann Condensation Coupling Reaction in the Formation of Aryl-Oxygen (C-O) Bonding by Copper-Mediated Catalyst

Garnier Color Naturals farba na vlasy CN 4 stredne hnedá 1x1 ks - Farby na vlasy, Starostlivosť o vlasy, Drogéria, kozmetika
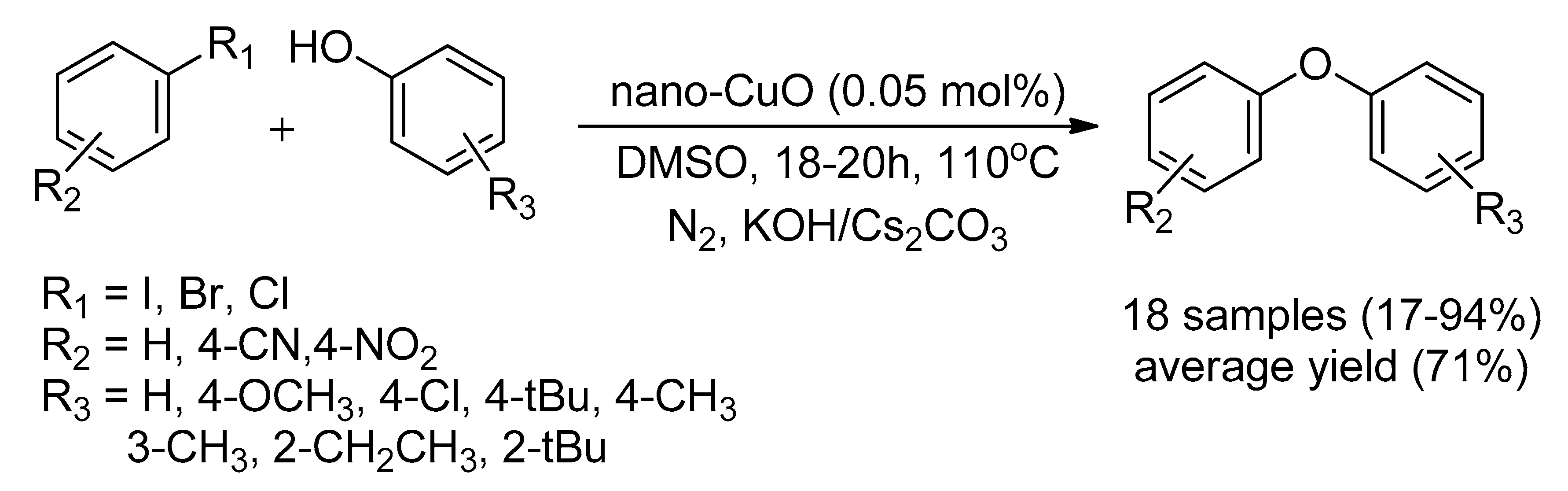
Catalysts | Free Full-Text | Recent Advancement of Ullmann Condensation Coupling Reaction in the Formation of Aryl-Oxygen (C-O) Bonding by Copper-Mediated Catalyst

Organic Electron Donors as Powerful Single‐Electron Reducing Agents in Organic Synthesis - Broggi - 2014 - Angewandte Chemie International Edition - Wiley Online Library















